Figure 3. (a) Ambiguous Necker cube
and (b) disambiguated cube variants.
During prolonged observation of the ambiguous Necker cube (Fig.
3a, Necker 1832), one of the most prominent ambiguous figures,
our perception becomes unstable and reverses spontaneously
between two possible interpretations (Fig. 3b). Adding tiny
depth cues disambiguates the Necker cube and stabilizes our
perception. In a series of EEG studies with very different
ambiguous stimuli and disambiguated stimulus variants we studied
differences between unstable and stable neural
representations (Kornmeier & Bach 2009, Kornmeier et al. 2016,
Joos 2020a and Joos 2020b). To our great surprise we a found
clear EEG-pattern with small amplitudes of two event-related
potentials (ERPs, P200 and P400) in the case of ambiguous
stimuli and large amplitudes in the case of disambiguated
stimulus variants (Fig. 4, columns 1 – 3).
However, stimulus ambiguity seems not to be the decisive factor,
because we found in the meanwhile very similar results using
stimuli with varying visibility (e.g. the mouth curvature of
happy and sad smiley faces (Fig. 4, left column) or stimuli
embedded in noise.
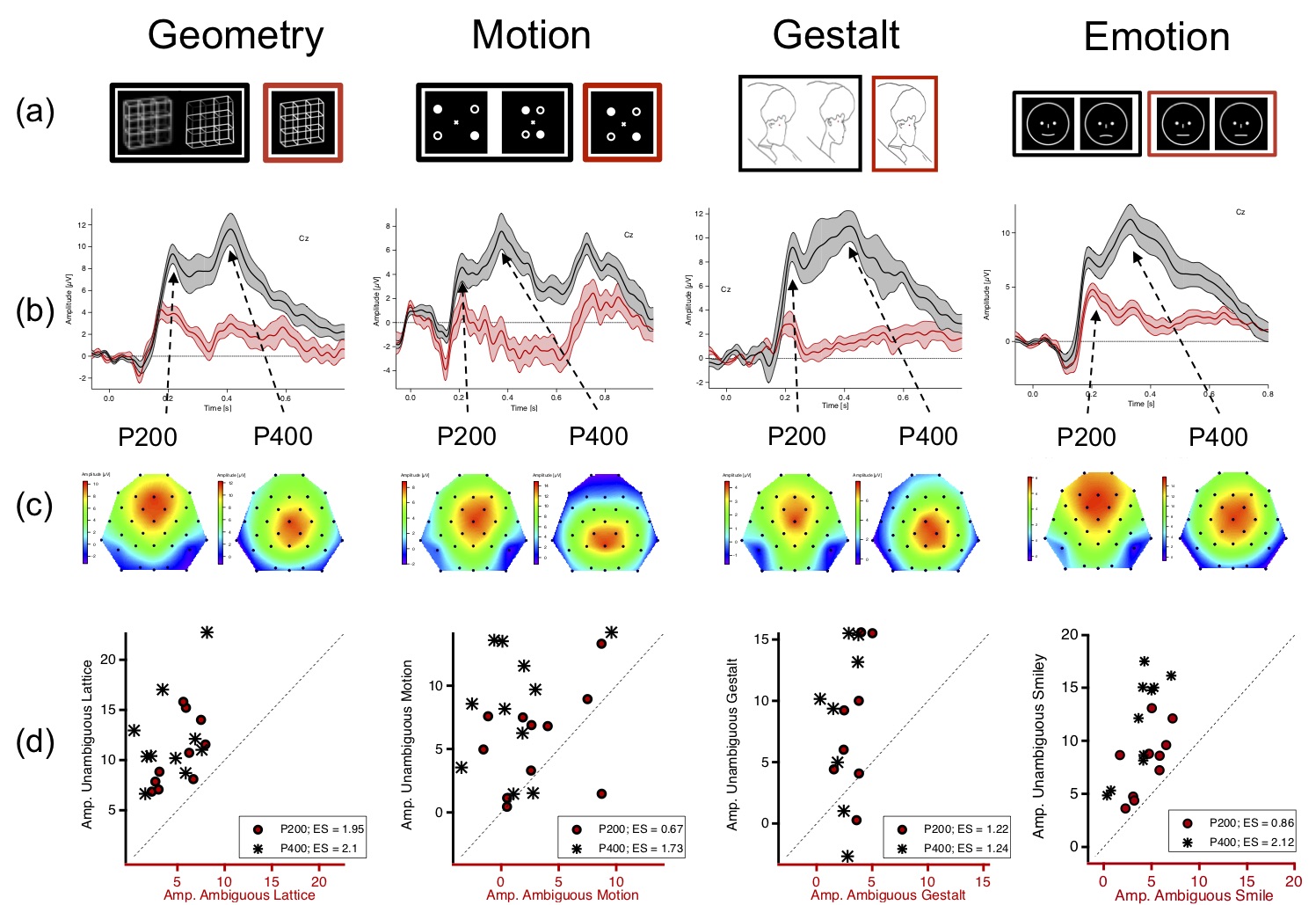
Figure 4. (a) From left to right: Necker lattice (geometry),
schematic presentation of the so-called motion quartett (Motion,
Von Schiller 1933; online Animation),
Boring's Old/Young Woman (Gestalt, Boring 1930), Smiley stimuli.
Black/red framed stimuli represent ambiguous/disambiguated
stimulus variants. (b) Grand Mean ERP traces (central traces) ± SEM
(above and below traces) from EEG elektrode Cz evoked by
ambiguous (red traces) or disambiguated (black traces) stimulus
variants. The disambiguated stimulus variants evoked significantly
larger ERP amplitudes. (c) Voltage maps for the P200 and P400 show
in pseudo-colors the distributions of the two ERP components
at certain time points (left/right: 200 and 400 ms after stimulus onset).
Red/blue colors indicate large/small amplitudes. Remarkable
is the similarity of the ERP results across very different
stimulus categories (columns). (d) Scatterplots representing
individual amplitude values of the different participants
(circles/stars represent P200 and P400 amplitudes). The large
amplitude differences are also clearly visible on the level
of individual participants.
In cooperation with the Department of Psychiatry and
Psychotherapy at the Medical Center of the University of
Freiburg and the Psychiatric Hospital Strasbourg, France we
investigate in this context patients with psychiatric disorders,
who also show altered perceptual and conscious states, in order
to test our hypotheses and the underlying models. Concurrently,
we also try to better understand the focused disorders.
Referenzen
Boring EG (1930). A new ambiguous figure.
Am J Psychol. 42, 444–445.
Joos E, Giersch A, Hecker L, Schipp J, Tebartz van Elst L &
Kornmeier J (2020a). Large EEG amplitude effects are
highly similar across Necker cube, smiley, and abstract
stimuli.
PLoS ONE 15(5): e0232928.
Joos E, Giersch A, Bhatia K, Heinrich SP, Tebartz van Elst L,
Kornmeier J (2020b) Using the perceptual past to predict the
perceptual future influences the perceived present – a novel ERP
paradigm PLoS
ONE 15(9): e0237663.
Kornmeier J & Bach M (2009). Object perception: when our brain
is impressed but we do not notice it.
Journal of Vision, 9(1),7 1-10.
Kornmeier J, Wörner R & Bach M (2016). Can I trust in what I
see? – EEG Evidencefor a Cognitive Evaluation
of Perceptual Constructs.
Psychophysiology, 53, 1507–1523.
Necker LA (1832). Observations on some remarkable optical
phaenomena seen in Switzer-land; and on an optical phaenomenon
which occurs on viewing a figure of a crystal or geo-metrical
solid.
The London and Edinburgh Philosophical Magazine and
Journal of Science, 1(5), 329–337.
Schiller PV (1933). Stroboskopische Alternativversuche.
Psychologische Forschung, 17, 179–214.